
Combining black soldier fly meal and
house fly larvae meal for a superior fish feed
09.12.2017 / Scienceandmore / Category: Animal Biology
Black soldier fly (BSF) and house fly (HF) have been established for the production of fish feed, and are approved for this purpose by EU legislation since July 2017. The larvae stage of both insects is mainly used since they are high in crude protein. The utilisation of black soldier fly larvae (BSFL) and house fly larvae (HFL) meal as replacement for fishmeal in fish feed has been investigated in several fish species and showed promising results.
Amino Acids
The nutrient content of BSFL and HFL has been shown to vary depending on their rearing substrate, but in general, they consist of 40 % to 60 % crude protein. Even though the larvae’s nutrient content can vary, their amino acid (AA) profiles stay constant. It is also important that the AA profile of fish feed is adjusted to the specific fish species and their nutritional needs. The fish species that are cultivated in aquacultures in the EU are mainly carnivorous and omnivorous, such as salmon and rainbow trout. They evolved with fish as significant nutrient source, and their metabolisms adapted to fish. Therefore, it is important that the AA profile of their feed matches the one of fishmeal. The AA profile of both BSFL and HFL is well balanced and, with some exceptions, relatively close to the one of fishmeal. However, their AA profiles could be optimised, which potentially would result in improved growth and development of fish in aquaculture when these meals are used as feeds. In order to achieve an optimisation, a combination of BSFL and HFL meal in equal amounts could compensate for lower amounts of particular amino acids in one of the meals. The AA profile of BSFL meal, HFL meal, and fishmeal are stated in table 1 (1).
Table 1: Amino acid composition (g/16 g nitrogen) of black soldier fly larvaemeal (BSFLM), house fly larvae meal (FHL), fishmeal, and a calculated combination of BSFLM and HFLM to equal parts.
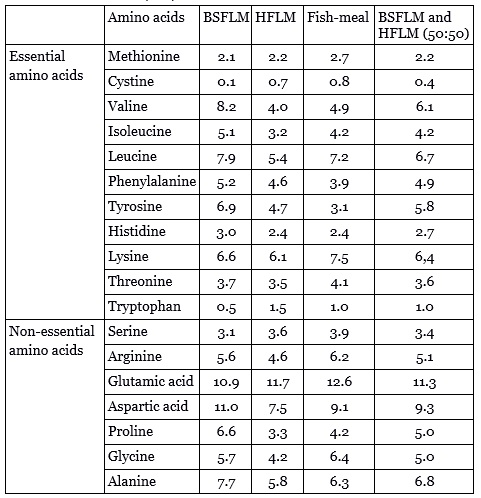
The low amounts of the essential AAs cysteine and tryptophan in BSFL could be compensated by the higher amounts in HFL and a combination would match the fishmeal proportion much closer. The high amounts of the non-essential AAs aspartic acid, proline and glycin in BSFL could compensate the lower amounts in HFL. Additionally, the proportions of the essential AAs valin, phenylalanine and tyrosine are higher in both insect species compared to fishmeal. However, both BSFL and HFL have lower amounts of lysine and threonine. These two AA could be supplemented to bring the AA profile of insect meal to the level of fishmeal. The other AA are similar in content compared to fishmeal (1,2).
The amino acid profile of soy, another substantial component of fish feed, is fairly similar to the one of fishmeal, too (1,2). This suggests that BSFL or HFL meal could possibly substitute for soy or fishmeal, or both. A reduction of soy and/or fishmeal in fish feed would decrease the environmental and ecological impact of these feed materials.
Calcium and phosphorous
BSFL are generally rich in calcium, whereas house fly larvae are very low (7.6 % and 0.5 % of dry mass). Here again, a combination of both insect species could compensate the lack in one species and raise the amount of calcium to fish meal levels (4.3 % of dry mass) (1,2). This, however, is of secondary concern since some fish species have been shown to obtain calcium directly from water (3). Phosphorous levels are low in both species, and an addition of phosphorous could potentially be required (1,2).
Fatty acids
Analyses of BSFL and HFL found that their lipid contents and fatty acid profile highly depend on their rearing substrate. Generally, the lipid content of BSFL is between 24 % and 49 %; and for HFL 9 % up to 26 %.
When BSFL and HFL were reared on cow manure, their fatty acid composition showed high levels of saturated and mono-unsaturated fatty acids, as well as low levels of omega-3 fatty acids, such as EPA and DHA. When fish offal was added to the cow manure, the content of beneficial omega-3 fatty acid in BSFL increased rapidly. Similarly, HFL that were reared on plant based substrate were low in omega-3 fatty acids (4,5).
The utilisation of BSFL and HFL as fish meal replacement in fish feed could therefore result in an alteration of the fatty acid profile in fish. Due to the inability of fish to produce omega-3 fatty acids themselves, they depend on the feed source for these fatty acids. It would be beneficial for fish performance, as well as for the consumer who eat these fish, if their feed would contain a higher amount of omega-3 fatty acids. Ultimately, fish are regarded as a source of omega-3 fatty acids by consumers. It was implicated that a diet with a low omega-6 to omega-3 fatty acid ratio (n-6:n-3 ratio), i.e. higher amounts of omega-3 fatty acids, leads to reduced inflammation and potentially improved prevention of cardiovascular heart disease (6).
The process of providing high amounts of omega-3 fatty acids to fish via larvae is complicated and not solved thus far. It has been suggested to feed larvae fish offal, microalgae and phytoplankton as omega-3 fatty acid sources. This, however, would be uneconomic since these materials could be fed to fish directly by mixing them in the fish feed (7). However, a specific process that is used in other livestock could be adapted. Here, the animals are fed a certain diet before their slaughter in order influence their fat content and fatty acid profile. Similarly, the fish rearing process could be separated in two stages. During the first stage, when the fish grow and develop rapidly, higher amounts of insect meal and insect oil instead of fishmeal and fish oil could be provided. During the second stage, before the fish are caught, higher amounts of fishmeal in the fish feed would increase their omega-3 fatty acid content. Ultimately, the aim is to achieve a high content of omega-3 fatty acids in fish for the consumer (4).
Overall, a combination of BSFL meal and HFL meal in fish feed could be superior to BSFL meal or HFL meal alone in terms of fish growth and development. However, future investigations are necessary to evaluate this approach.
References
1. Makkar, H.P.S., Tran, G., Heuzé, V., Ankers, P. (2014) State-of-the-art on use of insects as animal feed. Animal Feed Science and Technology, 197, 1-33.
2. Tran, G., Heuzé, V., Makkar, H.P.S. (2015) Inserts in fish diets. Animal Frontiers 5(2): 37-44.
3. Simkiss, K. (1974) Calcium metabolism of fish in relation to aging. In Aging of Fish (Ed. by Begenal, T.B., pp. 1-12. Unwin Brothers, Woking.
4. Henry, M., Gasco, L., Piccolo, G., Fountoulaki, E. (2015) Review on the use of insects in the diet of farmed fish: Past and future. Animal Feed Science and Technology, 203:1-22.
5. Hussein, M., Pillai, V. V., Goddard, J. M., Park, H. G., Kothapalli, K. S., Ross, D. A., et al. (2017) Sustainable production of housefly (Musca domestica) larvae as a protein-rich feed ingredient by utilizing cattle manure. PLoS ONE, 12(2), e0171708.
6. Simopoulos A.P. (2002) The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed Pharmacother. 2002 Oct;56(8):365-79.
7. Norambuena, F., Hermon, K., Skrzypczyk, V., Emery, J. A., Sharon, Y., Beard, A., & Turchini, G. M. (2015) Algae in Fish Feed: Performances and Fatty Acid Metabolism in Juvenile Atlantic Salmon. PLoS ONE, 10(4), e0124042.
